Background
Definition: Accumulation of blood in the pulmonary vasculature as a result of the inability of the left ventricle to pump blood forward adequately. Acute pulmonary edema, congestive heart failure and cardiogenic shock are a spectrum of diseases and should be considered and managed differently.
Epidemiology:
- 5 Million patients diagnosed with CHF in the US
- 500,000 new CHF diagnoses each year in the US
- Unclear what percentage of these patients will present with acute pulmonary edema (APE)
Causes: Acute myocardial infarction (AMI) is the most common cause of APE but there are a multitude of other causes including acute valvular pathology.
Pathophysiology: Our understanding of the pathophysiology of APE has changed dramatically over the last 70 years. The current model is based on the effects of neurohormones:
- Primary myocardial injury (AMI) or stress leads to decreased arterial blood pressure and renal perfusion
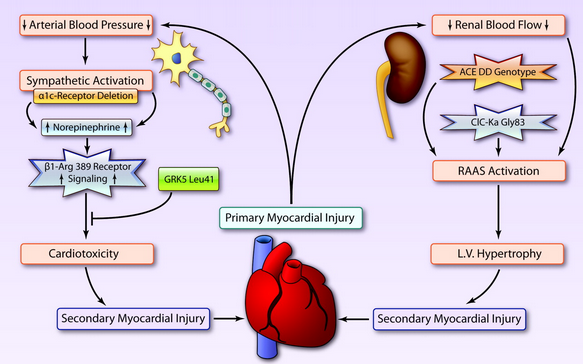
- Decreased arterial blood pressure causes sympathetic activation and release of neurohormones (i.e. norepinephrine).
- Decreased renal perfusion activates the renin-angiotensin-aldoserone system (RAAS)
- Increased circulating neurohormones cause peripheral vasoconstriction (increased afterload) and cardiotoxicity leading to secondary myocardial injury
- Splanchnic vasoconstriction leads to redistribution of blood contributing to increased preload and eventually, pulmonary volume overload
Symptoms
- Shortness of breath
- Dyspnea on exertion
- Diaphoresis
- Cough with pink sputum
- Chest pain
Signs
- Air hunger
- Hypoxia
- Tachycardia
- JVD
- Rales
- Skin pallor/mottling
- Altered Mental Status
- Decreased Urine Output
Immediate Management:
NB: Patients with APE have extremely tenuous respiratory status. As such, early management choices (first 10 minutes) determine whether these patients have good or bad outcomes.
Basics: ABCs, IV, O2, Cardiac Monitor, 12-lead EKG and POC Lung Ultrasound
Breathing
- Severe respiratory distress typically present and increased work of breathing can lead to fatigue as well as worsening cardiac function
- Apply non-invasive positive pressure ventilation (NIPPV)
- Multiple effects including decreasing work of breathing and stenting open alveoli during the entire respiratory cycle leading to improved gas exchange.
- NIPPV has been shown to reduce the need for intubation by decreasing work of breathing (Nava 2003, Bersten 1991)
- Limited evidence demonstrates an advantage of bilevel positive airway pressure (BPAP) over continuous positive airway pressure (CPAP) (Liesching 2014)
Circulation
- APE patients will have severely elevated blood pressures resulting from sympathetic activation and resultant vasoconstriction.
- Despite elevated blood pressures, end organ hypoperfusion occurs due to marked arterial vasoconstriction. This leads to acute kidney injury (AKI), intestinal ischemia, coronary ischemia and brain hypoperfusion.
- The lungs in a patient with APE are like an overflowing bathtub. We have to simultaneously stop the inflow of blood by turning off the tap (preload reduction) and increase outflow by unclogging the drain (afterload reduction)
12-Lead EKG
- Obtain an EKG as soon as possible to help identify etiologies of APE with specific indicated interventions.
- Myocardial ischemia and infarction are common causes of APE that EKG can rapidly identify.
- Life-threatening tachydysrhythmias may cause APE or occur due to ischemia.
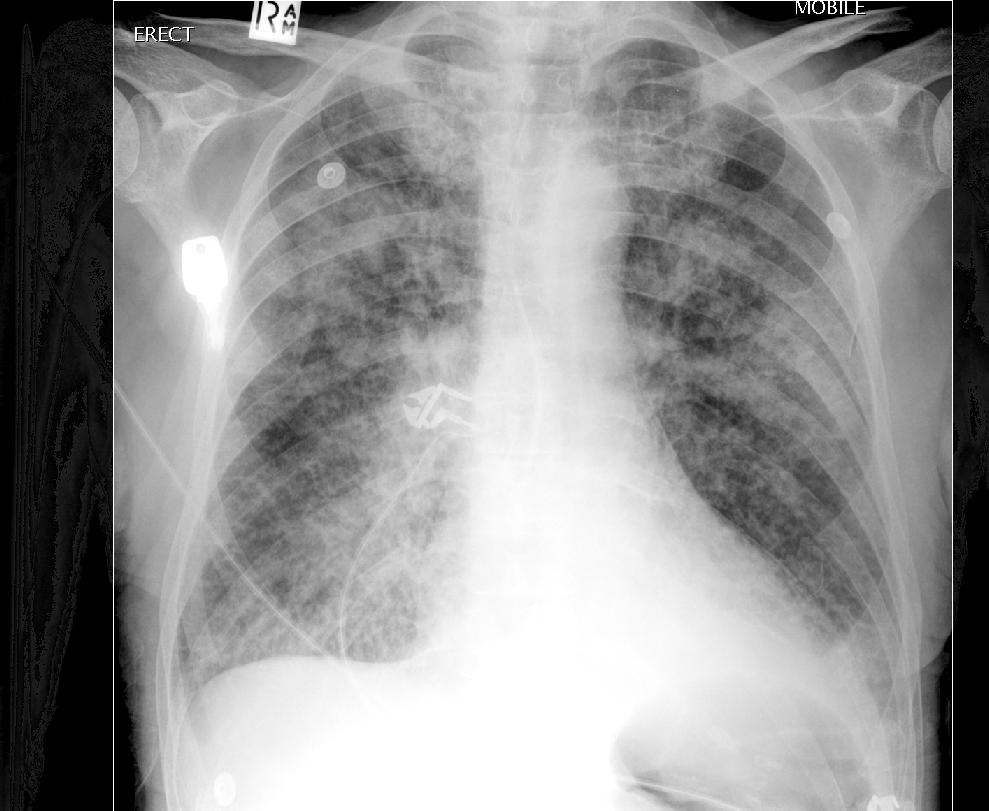
Chest X-Ray (CXR)
- May be helpful in confirming clinical diagnosis and in ruling out other possible etiologies.
- Most common finding: bilateral pulmonary congestion
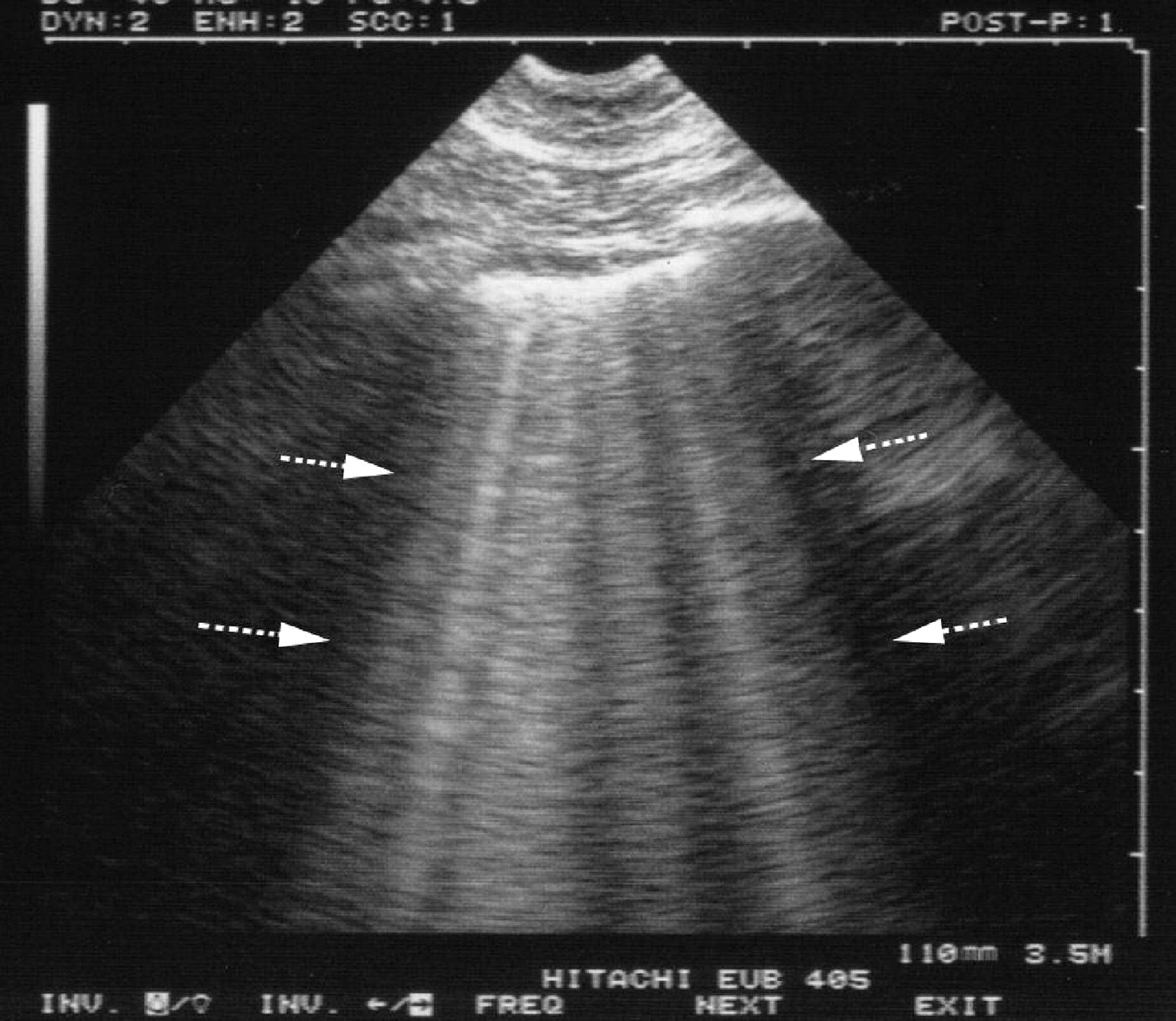
Point of Care Ultrasound (POCUS)
- Point of Care Ultrasound (POCUS)
- POCUS is an important diagnostic modality in patients with suspected APE
- In patients with APE, POCUS will demonstrate the presence of “B-lines”
- The presence of > 3 B-lines per rib space suggests the presence of interstitial pulmonary fluid.
- Read More: Lichtenstein’s BLUE Protocol
- Severe respiratory distress can be caused by a number of etiologies
- Presenting symptoms and signs overlap
- Diagnoses may be difficult to differentiate clinically
- Alternate diagnoses: asthma/COPD exacerbation, pulmonary embolism, pneumothorax
- Evidence demonstrates that physicians more accurately identify pulmonary edema on lung US than with CXR (Martindale 2012).
- A recent RCT demonstrated superiority of lung US in determining the final diagnosis of a patient presenting with undifferentiated respiratory distress (Laursen et al. 2014).
- Additionally, POCUS may identify a ruptured valve causing the patient’s symptoms leading to an alternate management pathway (i.e. cardiovascular surgery for valve repair)
Read More: US Against the World: Ultrasound in Differentiating COPD from CHF (Boring EM)
Directed Treatment
- Nitroglycerin (NTG)

- Low dose nitrates (< 100 mcg/min): cause venodilation leading to decreased preload
- High dose nitrates (> 100 mcg/min): cause arterial dilation reducing afterload
- Can be given sublingual while IV access is being obtained (Bussman 1978)
- If patient tolerates sublingual well, can start IV dosing at 50-75 mcg/min and titrate up rapidly
- Angiotensin Converting Enzyme Inhibitor (ACEI)
- Proposed mechanism: Interrupting the RAAS system leading to decreased neurohormonal drive. Leads to decreased afterload.
- Limited evidence demonstrates increased patient respiratory comfort and non-statistically significant decreases in respiratory failure (Hamilton 1996).
- ACEI are often unnecessary after aggressive NTG dosing
Less Useful Treatments
- Morphine
- Classic teaching from medical school endorses treatment of APE with “MONA” – Morphine, Oxygen, Nitroglycerin and Aspirin.
- Retrospective analysis of the Acute Decompensated Heart Failure Registry (ADHERE) database demonstrated an association between the use of morphine and increased mortality and ICU admission rate. (Peacock 2008)
- Read More: Morphine Kills in Acute Decompensated Heart Failure (REBEL EM)
- Loop Diuretics (i.e. furosemide)
- More than 50% of patients presenting in APE do not have volume overload but rather have volume redistribution (Zile 2008, Chaudhry 2007, Fallick 2011).
- Additionally, many patients with APE and volume overload will also have ESRD making loop diuretics noneffective in eliminating volume.
- Loop diuretics decrease glomeluar filtration rate (GFR), activate the RAAS, decrease cardiac output and increase afterload early after administration (Marik 2012).
Read More: Furosemide in the Treatment of Acute Pulmonary Edema (emDocs.net)
Take Home Points
- APE, CHF exacerbation and cardiogenic shock are different diseases and must be approached and treated differently.
- In patients presenting with undifferentiated respiratory distress, POCUS may be extremely helpful in rapidly determining the underlying disease.
- NIPPV should be rapidly applied to patients to support oxygenation and ventilation.
- Start nitrates early and rapidly titrate up to reduce both preload and afterload.
References:
Nava S et al. Noninvasive ventilation in cardiogenic pulmonary edema – a multicenter randomized trial. Am J Resp Crit Care Med 2003; 168: 1432-7. PMID: 12958051
Bersten AD et al. Treatment of severe cardiogenic pulmonary edema with continuous positive airway pressure delivered by face mask. NEJM 1991; 325 (26): 1825-30. PMID: 1961221
Liesching T et al. Randomized trial of bilevel versus continuous positive airway pressure for acute pulmonary edema. J Emerg Med 2014; 46(1): 130-40. PMID: 24071031
Lichtenstein DA, Meziere GA. Relevance of lung ultrasound in the diagnosis of acute respiratory failure: The BLUE protocol. Chest 2008; 134: 117-25. PMID: 18403664
Martindale JL et al. Diagnosing pulmonary edema: lung ultrasound versus chest radiography. Eur J Emerge Med 2012. PMID: 23263648
Laursen CB et al. Point-of-care ultrasonography in patients admitted with respiratory symptoms: a single-blind, randomised controlled trial. Lancet Respir Med 2014; 2: 638-46. PMID: 24998674
Bussmann W, Schupp D. Effect of sublingual nitroglycerin in emergency treatment of severe pulmonary edema. Am J Card 1978; 41: 931-936. PMID: 417614
Hamilton RJ et al. Rapid Improvement of acute pulmonary edema with sublingual captopril. Acad Emerg Med 1996; 3: 205-12. PMID: 8673775
Haude M et al. Sublingual administration of captopril versus nitroglycerin in patients with severe congestive heart failure. Intl J Card 1990; 27: 351-9. PMID: 2112516
Peacock WF et al. Morphine and Outcomes in Acute Decompensated Heart Failure: An ADHERE Analysis. Emerg Med J 2008; 25: 205 – 209. PMID: 18356349
Zile MR et al. Transition from chronic compensated to acute decompensated heart failure: pathophysiological insights obtained from continuous monitoring of intracardiac pressures. Circulation 2008; 118: 1433-41. PMID: 18794390
Chaudhry S et al. Patterns of weight change preceding hospitalization for heart failure. Circulation 2007;116:1549 –54. PMID: 17846286
Fallick C et al. Sympathetically mediated changes in capacitance: redistribution of the venous reservoir as a cause of decompensation. Circ Heart Fail 2011; 4: 669-75. PMID: 21934091
Marik PE, Flemmer M. Narrative review: the management of acute decompensated heart failure. J Intensive Care Med 2012; 27: 343-53. PMID: 21616957