Definition
- Targeted Temperature Management (TTM), also referred to as Therapeutic Hypothermia, is the maintenance of specific body temperature goals after return of spontaneous circulation (ROSC) post-cardiac arrest to reduce brain injury.
Mechanism
- Post ROSC, perfusion mismatch in the brain (decreased flow and increased metabolic demand) along with poor cerebral autoregulation, leads to permanent brain injury
- TTM decreases brain metabolic demand, cytokine release, free radials, ICP, and seizure activity (Polderman), thus protecting brain tissue from ischemic and reperfusion injury
- Early evidence exists demonstrating that TTM is also protective of cardiac tissue (Kang)
Evidence
- Two independent RCT’s in 2002 demonstrated that cooling patients to 32-34C after cardiac arrest improved neurological outcomes after out of hospital cardiac arrest due to VT/VF (OOHCA) (HACA, Bernard)
- Nielsen (2013) showed that cooling to 33C or 36C after OOHCA from VT/VF had similar survival and neurological outcomes
- Cochrane review (Arrich 2016) showed moderate evidence that TTM both improved survival outcomes (30% increase) and neurological outcomes (1.9 times more likely for favorable neurological status)
- The HYPERION trial (Lascarrou 2019) shows benefit for TTM in OOHCA due to non-shockable rhythms
- We recommend TTM for ALL cases of OOCHA w/ ROSC REGARDLESS of initial rhythm based on current best evidence
Candidates for TTM
- Individual hospitals often have specific guidelines on inclusion and exclusion criteria for their TTM protocols, however in general:
- Inclusion: patients post-cardiac arrest (requiring chest compressions or defibrillation) AND cannot follow commands after cardiac arrest (GCS Motor <6)
- Relative contraindications include – pregnancy, traumatic arrest or active bleeding, uncontrolled arrhythmia or hypotension, pregnancy, severe sepsis, and terminal illness with <6 months expected mortality, poor baseline mental status, DNR
Stage of TTM
- Initiation – cool target temperature (33 vs 36C depending on institution) ASAP (or within 4-6 hours post ROSC)
- Maintenance – stay at target temperature for 24 hours
- Rewarming – Gradually increase core temperature to normothermia (36 to 37.9C)
- Fever prevention – maintain normothermia for 72 hours
Initiation Methods (for the ED Physician)
- surface cooling: includes ice, cooling blankets, commercial cooling devices (ex. Arctic Sun)
- intravascular cooling catheters
- internal devices and devices with temperature feedback have improved outcomes (Calabro)
- administration of cold IV fluids is less favorable (Kim 2014)
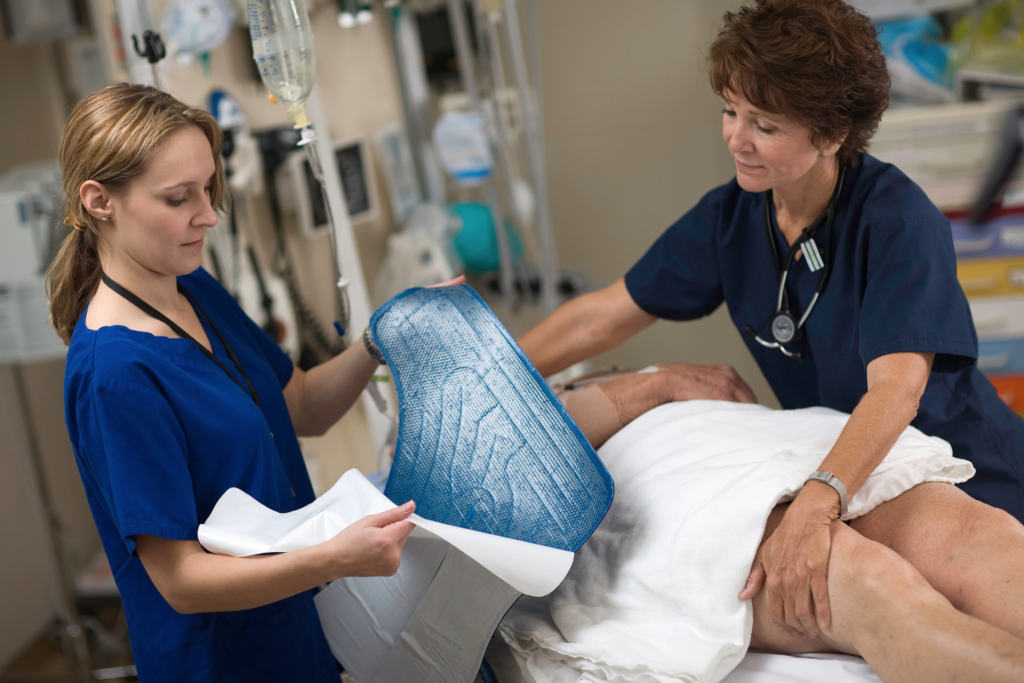
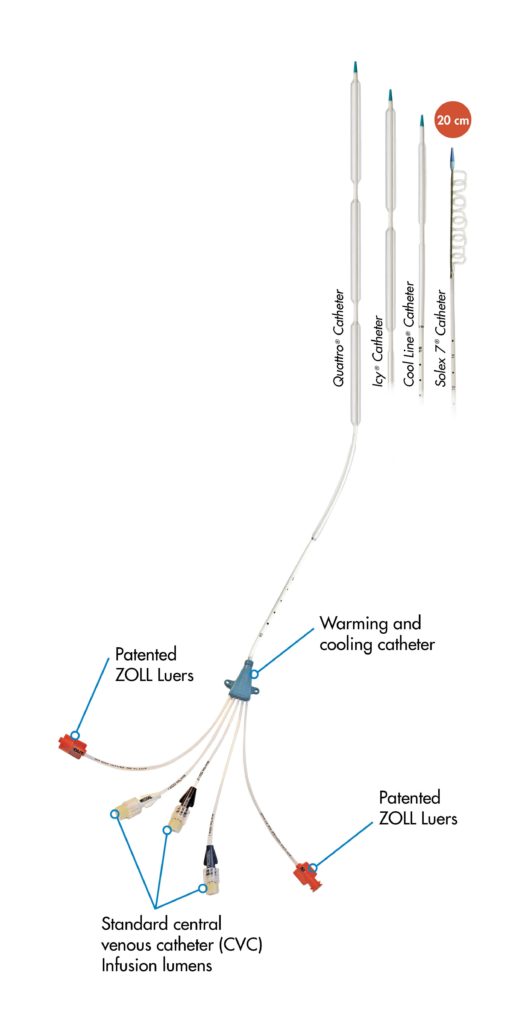
Peri-Cooling Adjuncts
- securing airway
- central venous access, arterial line
- baseline labs
- internal temperature probe (esophageal, bladder, or rectal)
- cooling DOES NOT prevent additional Post-ROSC workups, including CT, EKG, TTE, and even cardiac catheterization
Complications of TTM
- hypotension and bradycardia/arrhythmias: may require rewarming, pacing +/- pressors
- decreased O2 requirement from decreased metabolism: adjust vent for SpO2 95% and normo-carbia
- cold diuresis: modest increase in urine output during TTM induction – monitors I & Os and maintain euvolemia
- electrolyte shifts (HYPO Phos, Mag, Ca, K): replete accordingly (expect in rewarming process)
- hyperglycemia: due to decreased insulin production – monitor blood glucose and target goal 80-180; may require continuous insulin infusion, especially in patients with diabetes or glucose intolerance
- decreased GI motility: will require NG/OG tube (hold feeds during cooling)
- coagulopathy: stop TTM if there is uncontrolled bleeding
- decreased drug metabolism: consider with medication administration
Management of shivering
- shivering will start around 35C and paradoxically stop at 33C. Escalating therapies of management in order:
- skin counter warming
- standing Tylenol (to prevent fever bringing the temp back up into a range where shivering will occur as the cooling device tries to bring it back down)
- buspirone (30mg q8)
- IV magnesium (target serum level of 3-4)
- fentanyl or other opiates
- dexmedetomidine (do not use if bradycardic)
- propofol and benzodiazepines
- paralytics
Summary
- After successful ROSC, determine if your patient is a candidate for TTM based on inclusion and exclusion criteria
- If so, obtain labs with central venous and arterial access
- Initiate Cooling in the emergency department, including surface and/or internally
- Continue post-ROSC management including cardiac and neurological evaluations and interventions
- Consider complications including hypoxemia, electrolyte/glucose changes, and shivering and manage accordingly
- Follow your specific institution’s guidelines if available
Read more
- Rebel EM Podcast
- Sample Protocol by MD Anderson and UPenn
References
- Arrich J, Holzer M, Havel C, Müllner M, Herkner H. Hypothermia for neuroprotection in adults after cardiopulmonary resuscitation. Cochrane Database of Systematic Reviews 2016, Issue 2. Art. No.: CD004128. DOI: 10.1002/14651858.CD004128.pub4. Accessed 01 May 2021.
- Calabró, L., Bougouin, W., Cariou, A. et al. Effect of different methods of cooling for targeted temperature management on outcome after cardiac arrest: a systematic review and meta-analysis. Crit Care 23, 285 (2019). https://doi.org/10.1186/s13054-019-2567-6
- Bernard SA, Gray TW, Buist MD, Jones BM, Silvester W, Gutteridge G, Smith K. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med. 2002 Feb 21;346(8):557-63. doi: 10.1056/NEJMoa003289. PMID: 11856794
- Hypothermia after cardiac arrest (HACA) study group. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Eng J Med 2002;346:549-56. PMID: 11856793
- Kang IS, Fumiaki I, Pyun WB. Therapeutic Hypothermia for Cardioprotection in Acute Myocardial Infarction. Yonsei Med J. 2016;57(2):291-297. doi:10.3349/ymj.2016.57.2.291
- Kim F, Nichol G, Maynard C, Hallstrom A, Kudenchuk PJ, Rea T, Copass MK, Carlbom D, Deem S, Longstreth WT Jr, Olsufka M, Cobb LA. Effect of prehospital induction of mild hypothermia on survival and neurological status among adults with cardiac arrest: a randomized clinical trial. JAMA. 2014 Jan 1;311(1):45-52. doi: 10.1001/jama.2013.282173. PMID: 24240712.
- Lascarrou JB, Merdji H, Le Gouge A, Colin G, Grillet G, Girardie P, Coupez E, Dequin PF, Cariou A, Boulain T, Brule N, Frat JP, Asfar P, Pichon N, Landais M, Plantefeve G, Quenot JP, Chakarian JC, Sirodot M, Legriel S, Letheulle J, Thevenin D, Desachy A, Delahaye A, Botoc V, Vimeux S, Martino F, Giraudeau B, Reignier J; CRICS-TRIGGERSEP Group. Targeted Temperature Management for Cardiac Arrest with Nonshockable Rhythm. N Engl J Med. 2019 Dec 12;381(24):2327-2337. doi: 10.1056/NEJMoa1906661. Epub 2019 Oct 2. PMID: 31577396.
- Nielsen N, Wetterslev J, Cronberg T, Erlinge D, Gasche Y, Hassager C, Horn J, Hovdenes J, Kjaergaard J, Kuiper M, Pellis T, Stammet P, Wanscher M, Wise MP, Åneman A, Al-Subaie N, Boesgaard S, Bro-Jeppesen J, Brunetti I, Bugge JF, Hingston CD, Juffermans NP, Koopmans M, Køber L, Langørgen J, Lilja G, Møller JE, Rundgren M, Rylander C, Smid O, Werer C, Winkel P, Friberg H; TTM Trial Investigators. Targeted temperature management at 33°C versus 36°C after cardiac arrest. N Engl J Med. 2013 Dec 5;369(23):2197-206. doi: 10.1056/NEJMoa1310519. Epub 2013 Nov 17. PMID: 24237006.
- Polderman KH. Mechanisms of action, physiological effects, and complications of hypothermia. Crit Care Med. 2009 Jul;37(7 Suppl):S186-202. doi: 10.1097/CCM.0b013e3181aa5241. PMID: 19535947.
Images:
- https://www.bd.com/en-us/offerings/capabilities/patient-monitoring-and-temperature-management/targeted-temperature-management/arcticgel-pads
- https://www.zoll.com/products/temperature-management/catheter