Background
Acute Aortic Syndromes (AAS) are life threatening cardiovascular emergencies that are the bane of every emergency physician’s existence. They are diagnostic challenges due to the clinical presentation being highly non-specific. Computed tomography angiography (CTA), Transesophageal Echocardiography (TEE), and Magnetic Resonance Angiography (MRA) can help accurately diagnose AAS. CTA exposes patients to radiation and large doses of intravenous contrast, neither of which is a good enough reason to skip the test in patients you think may have a dissection, but certainly not something we want to do to all patients coming to the ED with chest pain. TEE and MRA may not be available or able to be performed in a timely manner. Having a clinical algorithm that can help physicians reduce misdiagnosis and at the same time avoid over-testing are lacking.
Clinical Question
Can the aortic dissection detection risk score (ADD-RS) be used to detect a low risk cohort for aortic dissection that can subsequently be safely “ruled out” with a negative D-dimer assay?
Population
Consecutive patients ≥18 years of age, with any 1 or more of the following symptoms, dating ≤14 days: chest/abdominal/back pain, syncope or perfusion deficit. All patients had to have acute aortic syndrome in the differential diagnosis
Intervention
Use of the ADD-RS of ≤1 + Negative D-Dimer (DD) for ruling out AAS
Outcomes
Primary: Failure rate of ADD-RS and D-dimer neg strategy for ruling out AAS
Calculation: AAS diagnoses/ number of patients with negative D-dimer within in a risk category
Secondary: Efficiency in ruling-out AAS
Calculation: Number of patients with negative D-dimer within a risk Category/number of enrolled patients
Design
Multicenter, prospective, observational study
Excluded
Primary trauma, unwillingness or inadequacy to participate in the study
Primary Results
Definitions:
- Pre-Test Probability Assessment (ADD-RS): Based on 12 risk markers classified in 3 categories
- Calculated by number of categories (0 – 3) where at least one risk marker was present
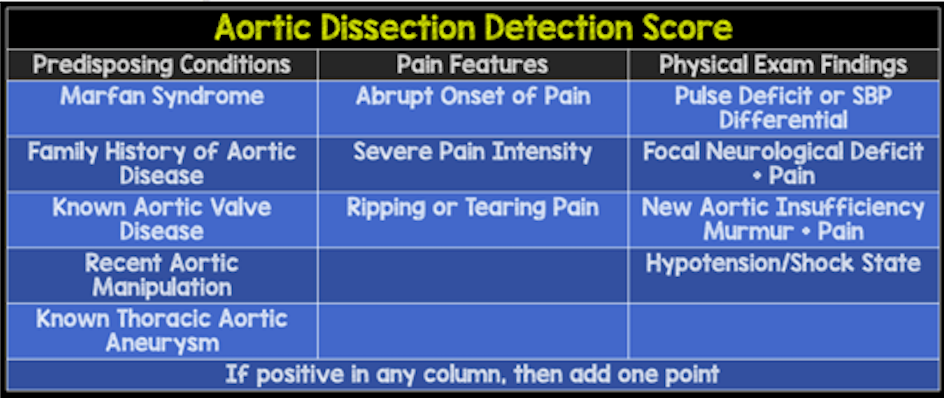
- D-Dimer: Defined as negative if <500ng/mL fibrinogen equivalent units (FEUs)
- Conclusive Imaging Used for Diagnosis of AAS: CTA, TEE, and/or MRA
Results:
- 1850 patients analyzed
- 438 (24%) patients had ADD-RS = 0
- 1071 (58%) patients had ADD-RS = 1
- 341 (18%) patients had ADD-RS > 1
- 241 (13%) patients had AAS
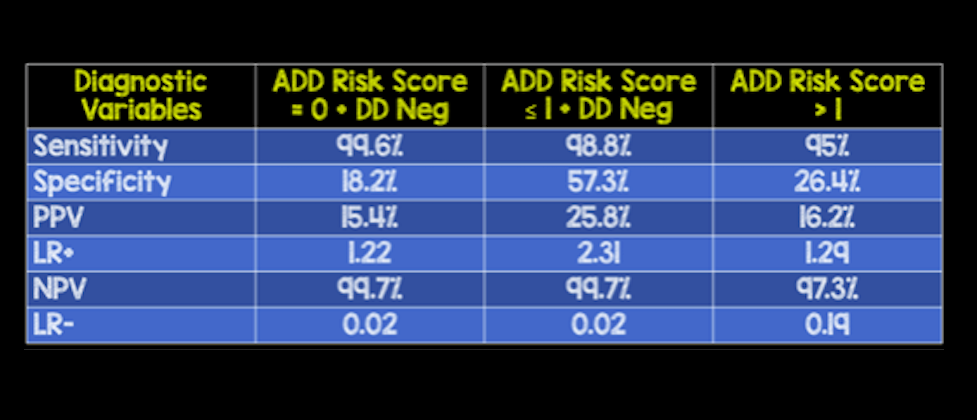
- Positive D-dimer (≥500 mg/mL) for Diagnosis for AAS
- 813 (43.9%) of patients
- 585 (38.8%) patients had ADD-RS≤1
- 228 (66.9%) patients had ADD-RS>1
- 8 patients with AAS had a negative D-dimer
- 813 (43.9%) of patients
Strengths
- Asks an important clinical question
- Multicenter study increases generalizability
- First study to really look at how to implement d-dimer into a diagnostic algorithm
- Addresses the call from ACEP clinical guideline to not incorporate d-dimer until a risk stratification tool had been developed
Limitations
- Observational study with many confounders
- Physicians not blinded to items for pre-test probability assessment or the D-dimer test results, potentially causing bias in this study
- Although symptoms triggering screening were pre-specified, entry criterion were provider determined
- Half the patients in this study did not have conclusive imaging and their case follow up was based on 14-day clinical follow up data only
- Unclear if 14 day follow up is an adequate time period
- There is no comparison to clinical gestalt
- Rate of AAD is pretty high (i.e. 13%), making it unclear how this would work in lower risk groups
- Only use one D-dimer assay, that may not be available at all institutions
- No discussion of age-adjustment of d-dimer
Other Issues
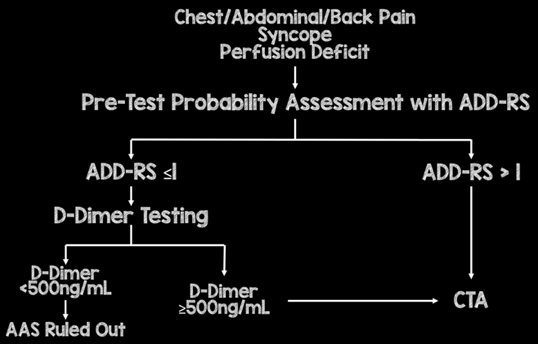
- In this paper, the authors suggest the following algorithm:
- ADD-RS>1, regardless of DD should proceed to CTA
- ADD-RS = 0 or ≤ 1 + DD neg are potentially ruled out for AAS
- This strategy will miss around 1 in 300 cases of AAS and avoid up to 1 in 2 CTA exams
Author's Conclusions
“Integration of ADD-RS (both = 0 or ≤ 1) with DD may be considered to standardize diagnostic rule-out of AAS.”
Our Conclusions
We agree with the authors conclusions with a stress on the “may be considered,” part of the conclusions.
Potential Impact To Current Practice
Further, prospective validation of this risk stratification system is needed before it can be widely applied in clinical practice.
Bottom Line
This is a novel clinical strategy in evaluating patients with the potential for aortic dissection, but still requires external validation, for reproducibility and comparison to overall clinical gestalt before implementation into clinical practice.
Read More
REBEL EM: The ADvISED Trial
Core EM: Aortic Dissection
