Background
Emergency Department (ED) patients frequently present with injuries and disease requiring procedural sedation and analgesia (PSA). PSA is a core skill for Emergency Physicians (EP) and acts to relieve pain and anxiety for patients. The procedure involves providing sedative and analgesic agents in order to allow EPs to effectively perform critical procedures from joint reduction to incision and drainage of abscesses to laceration repairs to preoxygenation.
Although PSA is commonplace in EDs, it is not without risks. Close monitoring is required to ensure patients do not have adverse events including, but not limited to hypotension, hypoxia, hypercarbia and loss of airway reflexes. A complete understanding of the magnitude of risk associated with PSA is difficult as small studies cannot establish safety particularly when adverse events are uncommon.
Clinical Question
What is the incidence of significant adverse events during PSA in the ED?
Design
Systematic review and meta-analysis
Literature Search
Comprehensive search of MEDLINE, EMBASE, EBSCO, CINAHL, CENTRAL, Cochrane Database of Systematic Reviews, Web of Science and Scopus from database creation to January 23, 2015 for original research including randomized control trials (RCT) and observational studies of PSA in adults > 18 years of age.
Outcomes
Estimated incidence per 1,000 patients with 95% CI for agitation, apnea, aspiration, bradycardia, bradypnea, hypotension, hypoxia, intubation, laryngospasm, and nausea/vomiting
Excluded
Studies of patients < 18 years of age. Studies that were not observational or RCTs.
Primary Results
Search strategy identified 2046 titles
- 55 articles, with 9,652 procedural sedations, were eligible for inclusion
- 25 were RCTs
- 30 were Observational studies
Critical Findings:
Most Frequent Events:
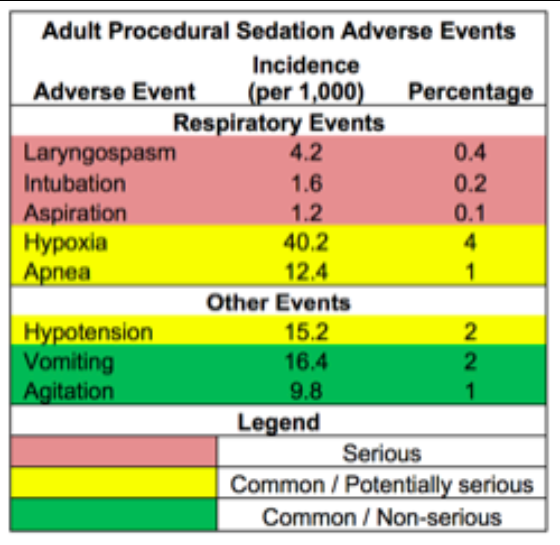
- Hypoxia: 40.2 per 1,000 sedations
- Vomiting: 16.4 per 1,000 sedations
- Hypotension: 15.2 per 1,000 sedations
- Apnea: 12.4 per 1,000 sedations
Severe Adverse Events Requiring Emergent Medical Intervention:
- Aspiration 1.2 per 1,000 sedations
- Laryngospasm: 4.2 per 1,000 sedations
- Intubation: 1.6 per 1,000 sedations
Additional Findings
- Table 1 – Incidence of Adverse Events per 1000 PSA
- Table 2 – Adverse Events by Medication Used for Sedation
Strengths
- Employed an expert librarian and performed and extensive search including 8 databases.
- Adhered to the Preferred Reporting Items for Systematic Reviews (PRISMA) Guidelines
- Used the Cochrane Collaboration bias appraisal tool to assess for bias in RCTs and the Newcastle Ottawa scale to asses for bias in cohort studies
- High level (kappa = 0.99) of interobserver agreement in study selection
- Most studies included had low to moderate risk of biases
Limitations
- Pediatric patients not included in the analysis so these results may not be applicable to patients < 18 years of age
- High statistical heterogeneity for following adverse events: hypoxia and agitation
- The authors were unable to determine what (if any) medications were administered prior to PSA. This could affect adverse events (i.e. opiates or benzodiazipines causing respiratory depression)
- Some of the outcomes (hypoxia, hypotension) had variable definitions in different studies, but were classified together as one outcome
- There was no discussion of what interventions were applied after adverse events
Author's Conclusions
“Serious adverse events during procedural sedation like laryngospasm, aspiration, and intubation are exceedingly rare. Quantitative risk estimates are provided to facilitate shared decisionmaking, risk communication, and informed consent.”
Our Conclusions
We agree with the author’s conclusions. Serious adverse events during ED PSA are extremely rare. This meta-analysis and systematic review gives us robust safety data, based on a large number of PSA events in the included studies.
Potential Impact To Current Practice
The information here can be used to reassure patients of the rarity of adverse events and be used to facilitate shared decision-making with patients (see pocket card from article supplement). Providers should remain diligent in PSA preparation and vigilant in monitoring for and managing adverse
Bottom Line
ED PSA is exquisitely safe. Serious adverse events like laryngospasm, aspiration and intubation rarely occur.
Read More
EMLit of Note: When Procedural Sedation Goes Wrong