 Background: Acetaminophen (aka APAP, paracetamol) has been used clinically since the 1950s and is one of the most widely used and trusted analgesic and antipyretic medications available. In addition to being available over the counter, it is present in numerous combination cold and pain medications. Over 130 million prescriptions were written for such compounds in 2010 alone. APAP overdose can be broadly divided into simple acute ingestion, wherein the evidence is strongest and the Rumack-Matthew nomogram is applicable, and cases with confounding factors, wherein expert opinion dictates practice.
Background: Acetaminophen (aka APAP, paracetamol) has been used clinically since the 1950s and is one of the most widely used and trusted analgesic and antipyretic medications available. In addition to being available over the counter, it is present in numerous combination cold and pain medications. Over 130 million prescriptions were written for such compounds in 2010 alone. APAP overdose can be broadly divided into simple acute ingestion, wherein the evidence is strongest and the Rumack-Matthew nomogram is applicable, and cases with confounding factors, wherein expert opinion dictates practice.
Epidemiology
- 50% of all acute liver failure cases in the United States are attributable to APAP
- ½ of these cases are intentional and ½ are unintentional (Larson 2005)
- Of those that are unintentional, 63% of cases occur in patients taking APAP-opioid combinations (Larson 2005)
- Mortality from acute liver failure secondary to APAP approaches 30% (Blieden 2014)
Clinical Presentation
- Key Point: The first stage of APAP toxicity is largely silent. Although there may be general malaise, nausea/vomiting, and abdominal pain, there may also be no symptoms (including normal LFTs).
- 24 hours: Transaminitis and possibly coagulopathy.
- 72 hours: Hepatic failure (jaundice, coagulopathy, encephalopathy, AKI)
- 96 hours: Survival or death
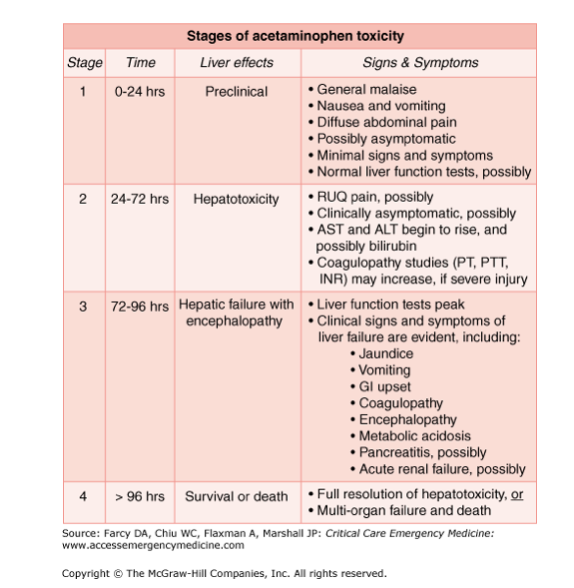
Stages of Acetaminophen Toxicity (Goldfrank’s Toxicology)
Differential Diagnosis
- The differential diagnosis can be broad depending on the time from ingestion to presentation and the symptoms at presentation
- Other causes of acute liver failure: alcoholic hepatitis, drug/toxin-induced hepatitis, viral hepatitis, shock liver, etc.
History of the Rumack-Matthew Nomogram:
- X-axis: Hours post-ingestion; Y-axis APAP concentration [APAP]
- Empirically derived by plotting untreated patients with acute APAP ingestion on the graph above and dividing those who developed hepatotoxicity from those who didn’t (defined as AST/ALT >1000). This approach led to a “300-line” from [APAP] =300 at 4 hours to [APAP]=50 at 12 hours (not pictured above) dividing patients with hepatotoxicity and those without.
- Threshold initially lowered to a “200-line” from [APAP]=200 at 4 hours to [APAP]=50 at 12 hours (dotted yellow line above) to be more conservative.
- With this threshold, only 60% of patients above the line would go on to develop hepatotoxicity.
- In the US, the line was further arbitrarily lowered 25% to current “150-line” from [APAP]=150 at 4 hours to [APAP]=37.5 at 12 hours (purple line above)
- With this cutoff, failure rate of the line in acute ingestions is ~1%. In other words, ~1% of patients below the line who are untreated will develop hepatotoxicity (defined as transaminitis)
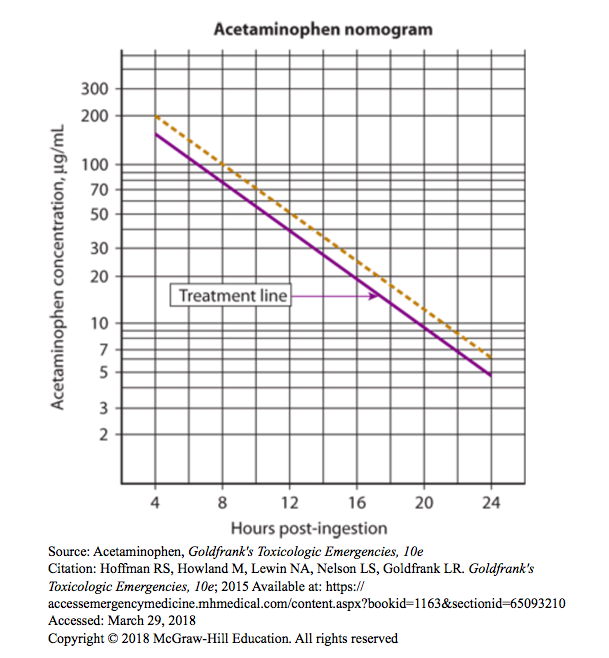
Acute APAP Ingestion
- Definition: Entire ingestion occurring within 8-hour period
- Measure APAP concentration at 4 hours from time of ingestion and plot on Rumack-Matthew Nomogram
- If [APAP] is above the 150-line, treat with NAC as below.
- If [APAP] is below the 150-line, there is no need to treat or gather further data points
- When time of ingestion is in doubt, use the earliest possible time of ingestion (similar to “last known well” in stroke care) as this provides the most conservative approach
- [APAP] prior to 4 hours after ingestion are not helpful and rather may be harmful by skewing management decisions (Seifert 2015). If patient arrives after 4 hours, get initial level and plot on nomogram above
- Treatment
- Supportive care
- ABCs:
- Airway management for somnolence
- IV fluids and pressors for hypotension
- Antiemetics for vomiting
- Transfusions prn (platelets/plasma for coagulopathy; pRBC if anemia from GIB)
- Lactulose for hepatic encephalopathy
- Hemodialysis for renal failure
- ABCs:
- Activated Charcoal
- Indicated if patient presents < 4 hours after APAP overdose
- Charcoal should only be administered in patients who are awake and able to take it orally OR have a protected airway and no contraindications
- Activated charcoal can decrease the probability of the 4-hour [APAP] being above the treatment line (Buckley 1999)
-
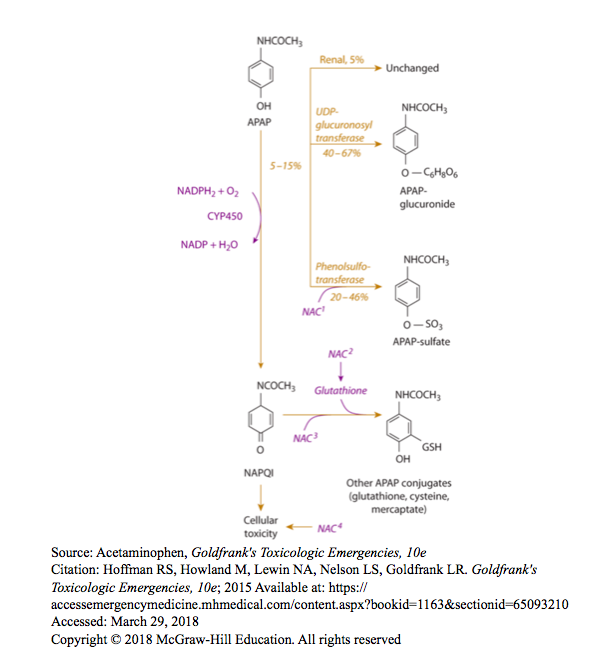
NAC Pharmacology (Goldfrank’s Toxicology)
N-acetyl cysteine (NAC)
- Mechanisms
- Prevents toxicity by increasing sulfation
- Detoxifies NAPQI by acting as both precursor and substitute for glutathione
- Treats toxicity at the cellular level (poorly understood)
- Treatment regimen
- IV and PO NAC are equally effective except in cases where liver failure is already present. Only IV NAC has been studied in liver failure cases
- IV associated with rare but severe anaphylactoid reactions
- Rate dependent
- Treatment: slow down bolus rate + give diphenhydramine
- DO NOT WITHHOLD NAC! If there is a concern for the degree of anaphylactoid reaction, switch to PO NAC instead
-
-
- Anaphylactoid reactions are more likely to occur in patients who have an undetectable APAP concentration (Schmidt 2013)
-
-
- Oral associated with >20% risk of emesis
- IV Protocol
- Loading dose: 150 mg/kg over 60 min, then
- 4 hour infusion 12.5 mg/kg/hr, then
- 16 hour infusion 6.25 mg/kg/hr
- PO Protocol
- Loading dose: 140 mg/kg, then
- 70 mg/kg q4hrs x 17 doses
- Variations from the classic regimen:
- Massive ingestion
- Generally defined as >50g
- Higher NAC doses (or extended treatment regiments) have been used with reduction in mortality (Chiew 2017)
- Massive ingestion
- Mechanisms
- Hemodialysis
- APAP acts as mitochondrial toxin when massive doses/very high concentration present
- Dialysis indicated in these situations
- NAC is dialyzable so NAC dose should be doubled during dialysis
- Fomepoizole
- New data showing usefulness due to inhibition of the CYP enzyme
- No standard care at this time
- Transplant
- Decision to proceed with liver transplant in acute liver failure 2/2 APAP overdose must take into account:
- Potential for spontaneous recovery (~70% of cases)
- Risk of major surgery in critically ill patient
- Psychosocial issues in intentional overdose and need for lifelong immunosuppression
- Multiple scoring criteria have been utilized to predict treatment failure/need for transplant/mortality.
- King’s College Criteria is the most validated and widely used model for prognostication (Bunchorntavakul 2018)
- Decision to proceed with liver transplant in acute liver failure 2/2 APAP overdose must take into account:
- Supportive care
More Complex Cases
- Inability to establish the time of ingestion
- Recommendation: measure the APAP concentration and AST, if either is elevated, treat with NAC
- Rationale
- AST elevation indicates evidence of hepatotoxicity
- Measurable APAP indicates potential for future toxicity
- Complications of NAC outweighed by potential benefits
- Acute ingestion of extended release APAP
- Concern for potential nomogram-crossing, i.e. initial APAP level below the treatment line but a subsequent level is above it
- Recommendation: Use the normal nomogram with a 4 hour level as for any other acute ingestion
- Rationale: Nomogram-crossing is thought to occur in up to10% of all acute APAP ingestions
- However, given that there may be a delayed peak [APAP], many still argue for measurement of a second [APAP] at 8 hours from time of ingestion (Bizovi 1996, Cetaruk 1997)
- Repeated supratherapeutic ingestions (AKA chronic overdose)
- Evidence-base
- 4g/day is safe
- Short term prospective studies show 6-8g/day is probably safe
- Patients with hepatoxicity have retrospectively reported mildly supratherapeutic dosing
- Theory: There may be some groups who could be at high risk of hepatotoxicity from repeated supratherapeutic ingestions (cytochrome p450 metabolism variation, variable glutathione stores, malnourishment)
- However, there continues to be no good evidence identifying groups that are unequivocally at higher risk (Caparrotta 2018)
- Recommendation: treat with NAC if:
- Ingestions were >200mg/kg/day
- People fall into “high risk” groups
- Evidence-base
- Pregnancy
- Concern exists because APAP does cross the placenta and fetal metabolism of APAP is not understood
- Recommendation: Use the nomogram as for non-pregnant patients
- Rationale:
- There are no reported cases of fetal or maternal toxicity in women with APAP concentrations below the 150-line
- There are no reported cases of fetal or maternal toxicity in women treated with NAC within 10 hours of ingestion, however some women in this group had elective abortions (Wilkes 2005).
- Increased time to NAC is associated with increased risk of spontaneous abortion (Wilkes 2005, Riggs 1989)
- Chronic heavy alcohol use
- Acute alcohol coingestion with APAP may be hepatoprotective (Waring 2008)
- Chronic heavy EtOH use may place someone at higher risk if they did NOT coingest EtOH with their APAP overdose (Bunchorntavakul 2018)
- Recommendation: use the nomogram as for those without a history of chronic heavy EtOH use
- Rationale: Patients with chronic heavy EtOH use were included in the derivation of the nomogram
Take home points
- APAP overdose is a common cause of acute liver failure in the United States and has significant morbidity/mortality
- APAP toxicity is often clinically silent in the first phase of overdose
- Evidence base is strongest for acute ingestions, in which the nomogram should be used to guide management with NAC
- Evidence is weaker and the clinical picture is more complex for unknown time of ingestion, extended-release APAP, IV APAP, chronic overdose, pregnancy, and in patients with chronic heavy EtOH use. In these circumstances, expert opinion guides management
- Always call your local poison control center in any patient with a suspected or known APAP overdose
Read More
LITFL: Paracetamol/Acetaminophen Overdose
MDCalc: Acetaminophen Overdose and NAC Dosing
UpToDate: Acetaminophen (paracetamol) poisoning in adults: Pathophysiology, presentation, and diagnosis
UpToDate: Acetaminophen (paracetamol) poisoning in adults: Treatment
Hendrickson RG. Acetaminophen. In: Hoffman RS, Howland M, Lewin NA, Nelson LS, Goldfrank LR. eds. Goldfrank’s Toxicologic Emergencies, 10e New York, NY: McGraw-Hill; 2015. Link
Tintinallis Emergency Medicine: a Comprehensive Study Guide, 8th ed. New York: McGraw-Hill Education; 2016. Chapter 190: Acetaminophen.
References
Beringer RM et al. Intravenous paracetamol overdose: two case reports and a change to national treatment guidelines. Arch Dis Child 2010;96(3):307-308. PMID: 21127004
Blieden M et al. A perspective on the epidemiology of acetaminophen exposure and toxicity in the United States. Expert Rev Clin Pharmacol. 2014;7(3):341-8. PMID: 24678654
Bizovi KE et al. Late Increase in Acetaminophen Concentration After Overdose of Tylenol Extended Relief. Ann Emerg Med. 1996;28(5):549-551. PMID: 8909277
Buckley NA et al. Activated charcoal reduces the need for N-acetylcysteine treatment after acetaminophen (paracetamol) overdose. J Toxicol Clin Toxicol. 1999;37(6):753-7. PMID: 10584587
Bunchorntavakul C, Reddy KR. Acetaminophen (APAP or N-Acetyl-p-Aminophenol) and Acute Liver Failure. Clinics in Liver Disease. 2018;22(2):325-346. PMID: 29605069
Caparrotta TM et al. Are some people at increased risk of paracetamol-induced liver injury? A critical review of the literature. Eur J Clin Pharmacol. 2017;74(2):147-160. PMID: 29067481
Cetaruk EW et al. Tylenol Extended Relief Overdose. Ann Emerg Med. 1997;30(1):104-108. PMID: 9209234
Chiew AL et al. Massive paracetamol overdose: an observational study of the effect of activated charcoal and increased acetylcysteine dose (ATOM-2). Clin Toxicol (Phila). 2017;55(10):1055-1065. PMID: 28644687
Larson AM et al. Acetaminophen-induced acute liver failure: results of a United States multicenter, prospective study. Hepatology. 2005;42(6):1364-72. PMID: 16317692
Riggs BS et al. Acute acetaminophen overdose during pregnancy. Obstet Gynecol. 1989;74(2):247. PMID: 2748061
Schmidt LE. Identification of patients at risk of anaphylactoid reactions to N-acetylcysteine in the treatment of paracetamol overdose. Clin Toxicol 2013; 51(6): 467-72. PMID: 23697458
Seifert SA et al. Acetaminophen concentrations prior to 4 hours of ingestion: Impact on diagnostic decision-making and treatment. Clin Toxicol (Phila). 2015;53(7):618-623. PMID: 26107627
Waring WS et al. Acute Ethanol Coingestion Confers a Lower Risk of Hepatotoxicity after Deliberate Acetaminophen Overdose. Acad Emerg Med. 2008;15(1):54-58. PMID: 18211314
Wilkes JM et al. Acetaminophen Overdose in Pregnancy. South Med J. 2005;98(11):1118-1122. PMID: 16351032
Woo OF et al. Shorter duration of oral N-Acetylcysteine therapy for acute acetaminophen overdose. Ann Emerg Med 2000; 35(4): 363-8. PMID: 10736123
